 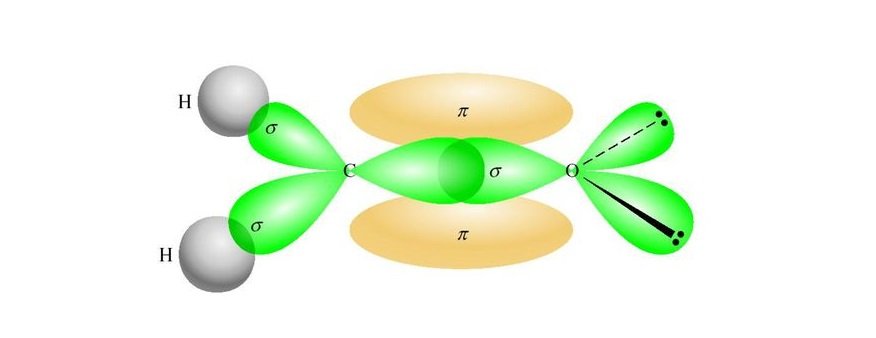
Hybrid orbitals (also sometimes referred to as group orbitals) are a way of simplifying the idea, by allowing us to combine an sp3 carbon, or an sp2 carbon, without having to always derive from first principles. It is, of course, worth remembering that these hand-drawn MO diagrams are just qualitative representations– true molecular orbitals are generated computationally using quantum mechanical calculations to approximate/calculate the various wave functions representing the MO's.įor more complex molecules (even something like ethene), it begins to get difficult to draw simple qualitative MO diagrams as there are so many orbitals to combine, and without computation, it can be difficult to know which orbitals to combine etc. In simple molecules (H-H, H-F etc.), simple molecular orbitals can be constructed on a piece of paper by combining atomic orbitals, to give the molecular orbitals. 
Nevertheless, I am sticking to the introductory, qualitative notions of VB and MO theory here. Wavefunctions exist in VB theory as well, except that they are constructed differently, and one might very well say that the simplistic MO theory above is, in fact, VB theory. 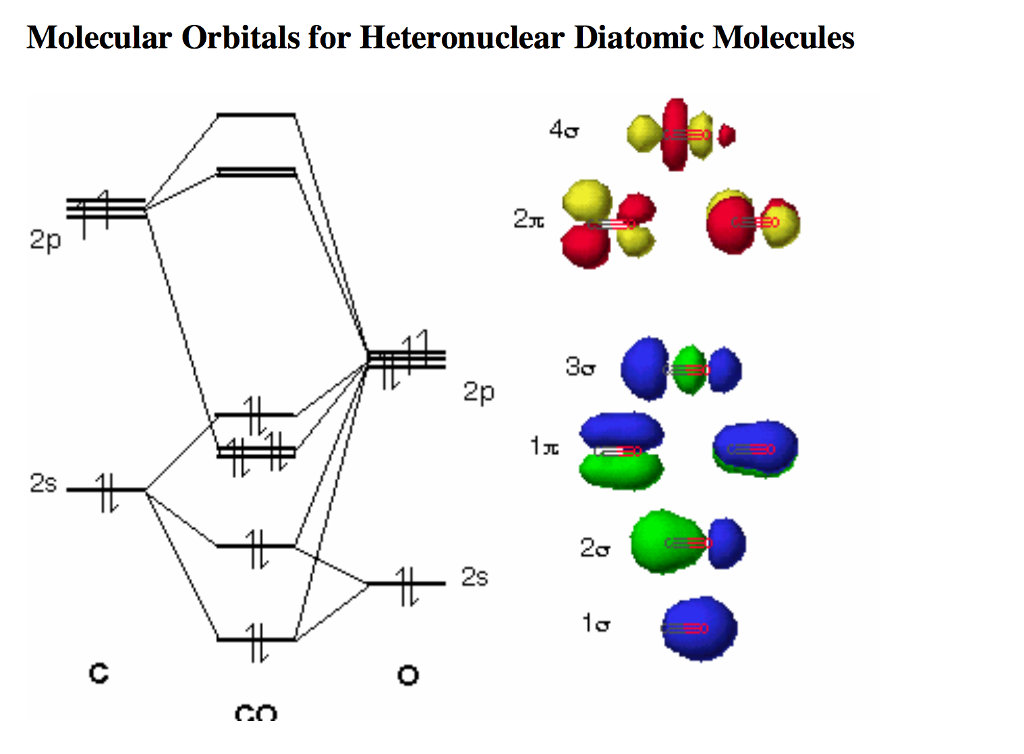 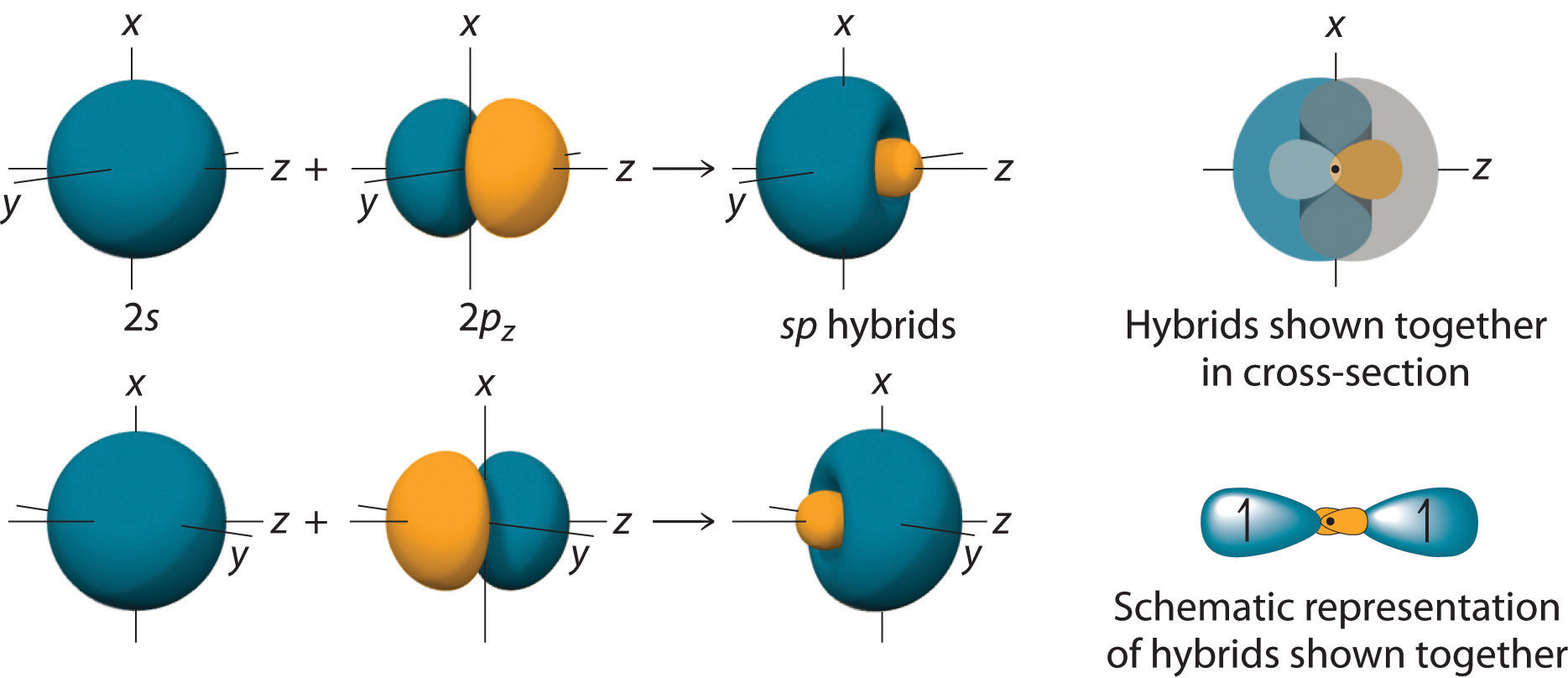
So, what you have done is: you have actually arrived at the same result (localised electron density) by another means (MOT).ĭisclaimer: There are people who will debate exactly what "VB" and "MO" theories are. But, remember that VB theory has no idea what an antibonding orbital is, or that it even exists. In terms of the electron density distribution, it is the same qualitative picture that is painted by VB theory. It's still LCAO-MO theory, just that the basis set of orbitals chosen is different, which leads to a different form of the wavefunction which happens to be localised between two nuclei. You didn't ask this question, but I anticipated you might wonder about it, since I've been going on and on about MOT only to end up saying that the electrons are localised. Then why do you still call it MO theory if the electrons are localised?!?!!?! Full delocalisation a la "proper" MO theory would imply that there are MOs which lead to bonding over the whole molecule, i.e. This is most obvious from the difference in the $\sigma$ orbitals shown above. One leads to a more accurate picture, but it becomes extremely tedious as the molecule becomes more complex.Īnd if hybrid orbitals do combine to form molecular orbitals, are the electrons still in between the two bonded atoms or are they delocalized like in the MO theory? You can do a little mix-and-match in order to simplify your life, without sacrificing too much accuracy.Īre molecular orbitals formed from regular (s,p,d, etc.) atomic orbitals, or are they also formed from the hybrid orbitals? The crux of the matter is, hybridisation can be applied to VB theory and it is actually in this context that you first meet hybridisation, but it is not exclusive to VB theory. What I wrote above mostly applies to this confusion. I was confused by this because I thought that VB and MO theories were two separate theories. Now that I've gotten that out of the way, I can proceed to answer your specific questions: Furthermore, guess which are the most important MOs that control the reactivity of ethene? Exactly it's the two $\pi$ orbitals. It's exactly the same as the $\pi_z$ MO in the real diagram! And the $\pi^*$ antibonding orbital in the simplistic diagram is also exactly the same as the $\pi_z^*$ orbital in the real diagram! This leads us to the main point: we can do this much simpler MO treatment using hybrid orbitals and get a sufficiently accurate MO picture, without having to go through a bunch of symmetry arguments to determine which AOs overlap and which AOs don't. However, the $\pi$ bonding orbital in your simplistic diagram is a resounding success. In fact, they are mostly there to provide the additional C-H bonding required to make the molecule stable, which is exactly what is neglected in the simplistic diagram. The other filled MOs are not likely to contribute since they are antibonding with respect to the two carbons. You could hybridise the $\mathrm$ orbital mixed in. Let's talk about a simple example, methane. The difference is that, instead of feeding the "pure" atomic orbitals into the LCAO mechanism, you carry out an additional mathematical step in order to get orbitals that have nice directional properties, and you feed those into the LCAO mechanism instead. It is still LCAO-MO theory, but just dumbed down a lot. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
